 Reactions of pyridines and primary amines with N-phosphorylated pyridines. The DMAP-catalyzed acetylation of alcohols–a mechanistic study. A single transition state in nucleophilic aromatic substitution: reaction of phenolate ions with 2-(4-nitrophenoxy)-4,6-dimethoxy-1,3,5-triazine in aqueous solution. Computational studies of nucleophilic substitution at carbonyl carbon: the S N2 mechanism versus the tetrahedral intermediate in organic synthesis. Ab initio study of the displacement reactions of chloride ion with formyl and acetyl chloride. 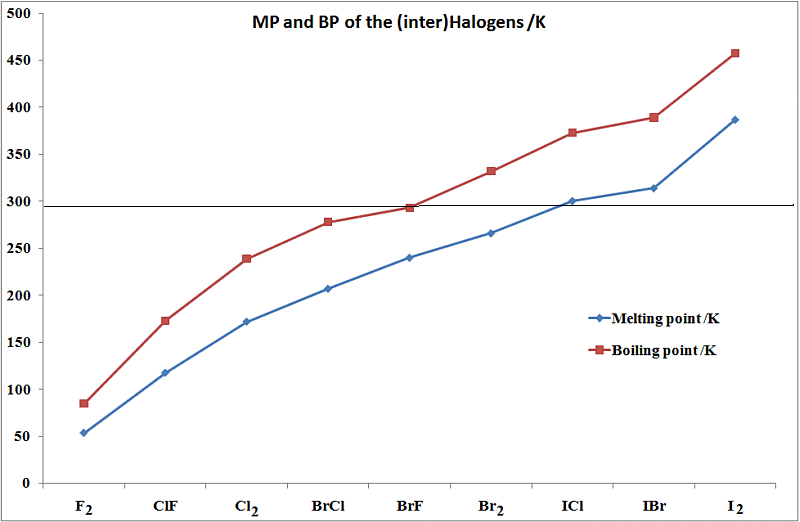
concerted or stepwise mechanisms for acyl transfer reactions of p-nitrophenyl acetate? transition state structures from isotope effects. Hydration of acylimidazoles: tetrahedral intermediates in acylimidazole hydrolysis and nucleophilic attack by imidazoles on esters: the question of concerted mechanisms for acyl transfer. Concerted mechanism for alcoholysis of esters: an examination of the requirements. Acyl-transfer reactions in the gas phase: the question of tetrahedral intermediates. Gas phase nucleophilic displacement reactions of negative ions with carbonyl compounds. Concerted acetyl group transfer between substituted phenolate ion nucleophiles: variation of transition state structure as a function of substituent. Concertedness in acyl group transfer: a single transition state in acetyl transfer between phenolate ion nucleophiles. A single transition state in the transfer of methoxycarbonyl group between isoquinoline and substituted pyridines. Structure activity studies on the equilibrium reaction between phenolate ions and 2-aryoxazolin-5-one-data consistent with a concerted acyl group transfer mechanism. Concerted mechanisms of acyl group transfer reactions in solution. retention of configuration for nucleophilic substitution at vinylic carbon. In-plane vinylic S N2 substitution and intramolecular β-elimination of β-alkylvinyl(chloro)-λ 3-iodanes. S N2 and Ad N-E mechanisms in bimolecular nucleophilic substitutions at vinyl carbon: the relevance of the LUMO symmetry of the electrophile. Is S N2 substitution with inversion of configuration at vinylic carbon feasible? J. An authentic case of in-plane nucleophilic vinylic substitution: the anionotropic rearrangement of di- tert-butyl thiirenium ions into thietium ions. Facile C–F bond formation through a concerted nucleophilic aromatic substitution mediated by the PhenoFluor reagent. Concerted nucleophilic aromatic substitution with 19F − and 18F −. Analysis of past and present synthetic methodologies on medicinal chemistry: where have all the new reactions gone? J. Rate and equilibrium studies in Jackson–Meisenheimer complexes. Modern Nucleophilic Aromatic Substitution (Wiley-VCH, Weinheim, 2013). Concerted Organic and Bio-Organic Mechanisms (CRC, Boca Raton, FL, 1999).   
This follows the reactivity trend for group 7 Halogens any halogen will displace halide ions from aqueous solution of any members of the group below it in the Periodic Table.Williams, A. Step 4 was repeated with bromine water and chlorine water as shown in the movie clip.Ĭolour changes due to displacement reactions were observed (are clearly visible in the movie clip).Ĭhlorine displaces both bromide and iodide ions from solution.īromine displaces iodide ions from solution. A few millilitres of aqueous iodine was added to one flask of potassium chloride, one flask of potassium bromide and one flask of potassium iodide and swirled.ĥ. 20ml potassium iodide was put into three 100ml conical flasks.Ĥ. 20ml potassium bromide was put into three 100ml conical flasks.ģ. Approximately 20ml potassium chloride was put into three 100ml conical flasks.Ģ. A series of free Science Lessons for 7th Grade and 8th Grade, KS3 and Checkpoint Science in preparation for GCSE and IGCSE Science.ġ. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
